science wire
Pharmacology
Results 3851 - 3900 of 4244.
Health - Pharmacology - 29.04.2014
Novartis gains FDA approval for Zykadia(TM), first therapy for patients with ALK+ NSCLC previously treated with the ALK inhibitor crizotinib
Zykadia (ceritinib) demonstrated an overall response rate of 54.6% in patients with ALK+ metastatic NSCLC who have no other treatment option Median duration of response to Zykadia was 7&period
Health - Pharmacology - 29.04.2014
CHMP recommends EU label expansion of Novartis’ Gilenya and data at AAN confirm efficacy on pre-treated MS patients
CHMP has recognized Gilenya's favorable benefit/risk profile and recommends EU label expansion to patients not responding to DMTs beyond interferon AAN data: Gilenya reduces relapse rates, new MRI lesion counts, brain volume loss & disability progression in pre-treated MS patients with high disease activity Gilenya is the only oral DMT that is effective across four key measures of MS (relapse rates, MRI lesions, brain volume loss and disabil
Health - Pharmacology - 25.04.2014
FDA Approves Roche’s HPV Test for First-Line Primary Screening for Cervical Cancer
FDA Approves Roche's HPV Test for First-Line Primary Screening for Cervical Cancer Expanded indication makes cobas HPV Test the only test approved in U.S. that can be used instead of Pap in first-line primary screening in women 25 and older Roche announced today that the U.S. Food and Drug Administration (FDA) approved the cobas HPV (Human Papillomavirus) Test for use as a first-line primary screening test for cervical cancer in women 25 and older.
Health - Pharmacology - 25.04.2014
Novartis’ INSTEAD study for Onbrez Breezhaler in patients with moderate COPD meets primary objective
Results confirm non-inferiority of Onbrez Breezhaler (indacaterol) in lung function compared to Seretide * (salmeterol/fluticasone) in the studied population In INSTEAD study, patients with moderate COPD and no exacerbations in the past year were switched from salmeterol/fluticasone to Onbrez Breezhaler Study showed similar symptomatic benefits in terms of shortness of breath and health status in patients treated with Onbrez Breezhaler compared
Economics - Pharmacology - 24.04.2014
Novartis delivered solid sales and profit growth in the first quarter of 2014
Net sales of USD 14.0 billion up 1% (+3% cc ) with all divisions contributing to growth (cc); core operating leverage (cc) driven by Pharmaceuticals and lower Corporate costs Operating income
Pharmacology - Health - 23.04.2014
New data at AAN to confirm efficacy of Novartis’ Gilenya across four key measures of MS disease activity, including brain volume loss
Gilenya reduced relapse rates, new MRI lesion counts, brain volume loss and disability progression in previously-treated MS patients with high disease activity Data at AAN showed significantly more Gilenya-treated patients (vs.
Economics - Pharmacology - 22.04.2014
Novartis announces portfolio transformation
Acquires GSK oncology products, strengthening Novartis' leading Oncology business with novel therapies and becomes GSK's preferred commercialization partner for its oncology pipeline Combines Novarti
Pharmacology - Economics - 09.04.2014
Novartis appoints Jeff George as Division Head, Alcon as Kevin Buehler retires; Richard Francis named as new Division Head, Sandoz
Basel, April 9, 2014 - Novartis announced today the appointment of Jeff George as Division Head of Alcon, effective May 1, 2014.
Health - Pharmacology - 07.04.2014
Novartis meningitis B vaccine Bexsero receives FDA Breakthrough Therapy designation in the US
The designation highlights the potential of Bexsero to meet the urgent need for a licensed vaccine in the US against unpredictable and devastating meningitis B Bexsero, already approved in Europe, Ca
Health - Pharmacology - 07.04.2014
Novartis and Malaria No More provide two million antimalarial treatments to children in Zambia
One million antimalarial treatments donated by Novartis, matching one million treatments funded through Malaria No More's Power of One campaign Power of One enables the public to fund malaria tests a
Pharmacology - Health - 03.04.2014
Novartis data on 19 compounds at AACR highlight strong cancer pipeline across multiple molecular targets and biological pathways
More than 50 abstracts highlight breadth and depth of Novartis Oncology pipeline and ability to test various combinations early to target multiple pathways Data show activity of investigational compo
Pharmacology - Health - 31.03.2014
PARADIGM-HF trial of Novartis’ LCZ696 for chronic heart failure closes early based on strength of interim results
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Pharmacology - Health - 27.03.2014
Novartis to work with FDA on path forward for RLX030 for acute heart failure following Advisory Committee outcome
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Pharmacology - Health - 26.03.2014
Study published in NEJM showed LDK378 demonstrated an overall response rate of 58% in patients with ALK+ NSCLC
Novartis study published in NEJM showed LDK378 demonstrated an overall response rate of 58% in patients with ALK+ NSCLC Data showed investigational treatment LDK378 (ceritinib) also achieved a median progression-free survival of seven months Patients with this type of lung cancer, especially those for whom prior therapies have failed, are in need of new treatment options Data served as the basis for regulatory application to the US Food and Drug
Pharmacology - Health - 22.03.2014
New phase III psoriasis data show rapid, significant skin clearance
New phase III psoriasis data show rapid, significant skin clearance and convenient administration with Novartis' secukinumab (AIN457) Pivotal phase III FEATURE and JUNCTURE studies met primary and pre-specified secondary endpoints, showing high efficacy with convenient pre-filled syringe (PFS) and autoinjector/pen (AI) , Patient-reported outcomes from FEATURE and JUNCTURE show high patient satisfaction with secukinumab PFS and AI self-administra
Pharmacology - Health - 21.03.2014
Novartis announces US FDA approval of Xolair for chronic idiopathic urticaria (CIU)
Report a suspected side effect (also known as an adverse event) related to a Novartis Pharmaceutical drug or a Novartis Vaccine.
Health - Pharmacology - 21.03.2014
Novartis announces start of new secukinumab (AIN457) versus Stelara (ustekinumab) phase IIIb head-to-head psoriasis study at AAD 2014
Novartis announces start of new secukinumab (AIN457) versus Stelara (ustekinumab) phase IIIb head-to-head psoriasis study at AAD 2014 Global phase IIIb head-to-head study of secukinumab versus Stelar
Pharmacology - Health - 20.03.2014
Novartis introduces Lucentis in state of the art pre-filled syringe
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Health - Pharmacology - 19.03.2014
Novartis reports data at EBCC-9 for secondary survival endpoint of Afinitor trial in HR+/HER2- advanced breast cancer
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Health - Pharmacology - 13.03.2014
Novartis Bexsero meningitis B vaccine receives clinical recommendation for use in infants and adolescents in Australia
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Pharmacology - Health - 07.03.2014
Novartis announces positive Phase III study for Jakavi in patients with rare blood cancer polycythemia vera
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Pharmacology - Health - 06.03.2014
Novartis announces Xolair approved in EU as first and only licensed therapy for chronic spontaneous urticaria (CSU) patients unresponsive to antihistamines
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Pharmacology - Health - 28.02.2014
Novartis marks Rare Disease Day with call for more research to understand and find treatments for rare diseases
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.m. 5:00 p.m. EST More information Global Media Relations Eric Althoff Basel, Switzerland +41 61 324 7999 eric.althoff [a] novartis (p) com More information Investor Relations Switzerland investor.relations [a] novartis (p) com
Pharmacology - Health - 26.02.2014
Novartis extends leadership in clinical trial data transparency, reinforcing its support of clinical research and innovation
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.m. 5:00 p.m. EST More information Global Media Relations Eric Althoff Basel, Switzerland +41 61 324 7999 eric.althoff [a] novartis (p) com More information Investor Relations Switzerland investor.relations [a] novartis (p) com
Economics - Pharmacology - 25.02.2014
Novartis shareholders approve all resolutions proposed by Novartis Board of Directors
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Pharmacology - Health - 21.02.2014
Novartis shows continued commitment in Japan with Lucentis approval in fourth Japanese indication, diabetic macular edema
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.m. 5:00 p.m. EST More information Global Media Relations Eric Althoff Basel, Switzerland +41 61 324 7999 eric.althoff [a] novartis (p) com More information Investor Relations Switzerland investor.relations [a] novartis (p) com
Health - Pharmacology - 19.02.2014
Novartis investigational compound LDE225 met primary endpoint in pivotal trial for patients with advanced basal cell carcinoma
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Pharmacology - Health - 17.02.2014
Novartis expands cancer immunotherapy research program with acquisition of CoStim
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Economics - Pharmacology - 29.01.2014
Novartis delivers strong sales and innovation in 2013; underlying business performance reinforces growth prospects
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Pharmacology - Economics - 27.01.2014
Novartis recognized among world’s most sustainable companies in Corporate Knights Global 100
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Pharmacology - Health - 24.01.2014
Novartis reports positive opinion from CHMP for Xolair in severe form of skin disease CSU, a debilitating form of hives and chronic itch
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Pharmacology - Health - 24.01.2014
Novartis to request re-examination of serelaxin (RLX030) in acute heart failure (AHF) for conditional marketing authorization in EU
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Pharmacology - Health - 12.01.2014
Hervé Hoppenot, President of Novartis Oncology, leaves Novartis to pursue career opportunity outside of organization
Main switchboard Switzerland +41 61 324 11 11 +41 61 324 80 01 Monday - Friday, 8:30 a.m. 5:00 p.m. GMT+1 US switchboard +1 862 778 21 00 Monday - Friday, 8:30 a.
Pharmacology - Health - 19.12.2013
Sandoz begins Phase III clinical trial for biosimilar adalimumab
Sandoz advances leading biosimilars pipeline by strengthening Immunology portfolio Global program marks Sandoz' sixth biosimilar molecule to enter Phase III testing Trial will support registration in
Health - Pharmacology - 18.12.2013
Sandoz receives first approval for AirFluSal Forspiro
Novel inhaler approved for patients with asthma and COPD Approval follows completion of EU decentralized procedure (DCP) AirFluSal Forspiro strengthens Sandoz respiratory portfolio and reinforces com
Pharmacology - Health - 09.12.2013
Novartis drug Jakavi improved overall survival of patients with myelofibrosis in four separate analyses of long-term Phase III studies
In two Phase III studies, Jakavi reduced the risk of death and maintained spleen reductions at three years compared to conventional therapy and placebo Similar survival benefit seen in patients with and without high-risk mutations Separate analysis shows Jakavi may increase the probability of 10-year survival of myelofibrosis patients by more than 50% compared to conventional therapy Jakavi is the only JAK inhibitor approved in more than 50 coun
Pharmacology - Health - 09.12.2013
New five-year data support superiority of Novartis drug Tasigna over Glivec in newly diagnosed Ph+ CML patients
ENESTnd data indicate trend for longer overall survival and event-free survival in newly diagnosed Ph+ CML patients on Tasigna versus Glivec Data demonstrated higher rates of early and deeper molecul
Health - Pharmacology - 07.12.2013
Novartis highlights research on investigational, personalized’T cell therapy CTL019 in patients with forms of acute and chronic leukemia
Data at ASH show increased scientific understanding of CTL019 and its potential role in the treatment of certain types of lymphocytic leukemia , , , Presentations include findings that 19 of 22 pediatric patients with acute lymphoblastic leukemia ( ALL) (86%) experienced complete remissions Novartis and Penn exclusive global collaboration to develop chimeric antigen receptor (CAR) technology is moving forward with the goal of expanding clinical
Pharmacology - Health - 06.12.2013
Novartis investigational compound LBH589 significantly extended time without disease progression in Phase III multiple myeloma study
Study of LBH589 plus bortezomib and dexamethasone met primary endpoint of extending PFS compared to bortezomib plus dexamethasone and placebo LBH589 has potential to be the first in its class of anticancer agents available to patients with multiple myeloma Data will be presented at an upcoming medical congress and discussed with regulatory authorities worldwide Basel, December 6, 2013 - Novartis today announced that results of a Phase III trial
Health - Pharmacology - 05.12.2013
Novartis Africa Day highlights company’s efforts to expand access to healthcare
Africa's healthcare challenges are compounded as it begins to face a dual disease burden as non-communicable diseases rise In addition to existing corporate responsibility efforts such as its Malaria
Pharmacology - Health - 04.12.2013
Novartis to highlight long-term data, innovative pipeline for patients with hematologic diseases and breast cancer at ASH and SABCS
Multiple studies evaluating deep molecular response with Tasigna versus Glivec in Ph+ CML patients Overall survival data from Jakavi Phase III trials in patients with the debilitating blood cancer, m
Pharmacology - Life Sciences - 04.12.2013
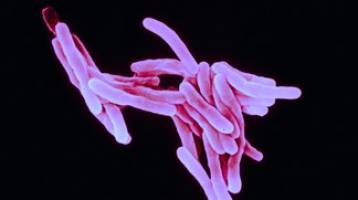
A natural antibiotic turns out to be a lethal weapon in the fight against tuberculosis. Scientists have discovered it has an unexpected dual action that dramatically reduces the probability that TB bacteria will become resistant. Technology has made it possible to synthesize increasingly targeted drugs.
Pharmacology - Economics - 22.11.2013
Novartis highlights growth prospects driven by significant R&D pipeline progress and the expected increase in blockbuster treatments
Novartis takes action to strengthen portfolio and capital allocation, starts share buyback Blood transfusion diagnostics unit divested as part of ongoing portfolio management Reiterates optimal capit
Life Sciences - Pharmacology - 17.11.2013
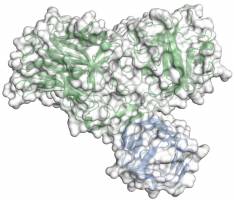
Media Releases Biology Research Using Synchrotron Light Human Health Researchers elucidate how botulinum neurotoxin A binds to its protein receptor and thus provide a basis for the development of new drugs Botulinum neurotoxin A, better known as botox, is a highly dangerous toxin that causes paralysis in man that may prove fatal.
Health - Pharmacology - 14.11.2013
Novartis honored by Malaria No More corporate citizenship award in recognition of long-standing commitment to eliminating the disease
Pharmacology - Innovation - 14.11.2013
Novartis announces positive clinical trial results for novel H7N9 vaccine
85% of subjects immunologically protected after receiving second dose of investigational cell culture vaccine when combined with proven MF59 adjuvant Vaccine now in large scale production highlightin
Pharmacology - Economics - 11.11.2013
Novartis announces divestiture of its blood transfusion diagnostics unit to Grifols
Health - Pharmacology - 23.10.2013
Alcon receives positive NICE recommendation for Jetrea to treat eligible patients with vitreomacular traction, including macular hole
One-time single eye injection of Jetrea recognized as clinically and cost-effective treatment for eligible patients with vitreomacular traction, including macular hole Jetrea is the first and only ap
Economics - Pharmacology - 22.10.2013
Novartis delivers strong sales performance in third quarter driven by growth products’ momentum; innovation newsflow reinforces growth prospects
Group net sales up 4% (+6% cc ) to USD 14.3 billion in third quarter and up 2% (+4% cc) in first nine months; all divisions contributed to growth Negative currency impact of 6 percentage points
Pharmacology - Health - 05.10.2013
Novartis announces positive results from final Phase III omalizumab registration study in severe form of chronic skin disease CSU
Omalizumab significantly reduced itch and hives caused by chronic spontaneous urticaria (CSU) as early as Week 1; benefit sustained over 24 weeks of active treatment Omalizumab 300 mg was nearly twice as effective in improving patients' quality of life within 12 weeks of treatment versus placebo ASTERIA I is the final omalizumab CSU registration study to be presented; regulatory applications were filed with EU and US authorities in Q3 2013 CSU i
Life Sciences - Mar 27
Understanding the Brain - TU Ilmenau's EU EMBRACE Project Nominated for European Excellence Award
Understanding the Brain - TU Ilmenau's EU EMBRACE Project Nominated for European Excellence Award
Social Sciences - Mar 27
A manual addresses, for the first time in Spain, child and adolescent sexual exploitation
A manual addresses, for the first time in Spain, child and adolescent sexual exploitation

Environment - Mar 26
Changing vegetation in thawing permafrost increases emissions of greenhouse gases
Changing vegetation in thawing permafrost increases emissions of greenhouse gases

Environment - Mar 26
University of Manchester hits major sustainability milestone, with Main Campus becoming 100% 'Zero Landfill'
University of Manchester hits major sustainability milestone, with Main Campus becoming 100% 'Zero Landfill'

Social Sciences - Mar 26
"It would be naive to believe that a social media ban will solve all problems"
"It would be naive to believe that a social media ban will solve all problems"

Health - Mar 26
Earlier detection, better outcomes: Irish researchers target rising bowel cancer rates with new blood test
Earlier detection, better outcomes: Irish researchers target rising bowel cancer rates with new blood test
Environment - Mar 26
UK must improve energy efficiency to end 50 years of policy failure and prevent future energy crises, study argues
UK must improve energy efficiency to end 50 years of policy failure and prevent future energy crises, study argues

Mathematics - Mar 26
From Materials to Medical Imaging, Fonseca's Work Shapes the Future of Innovation
From Materials to Medical Imaging, Fonseca's Work Shapes the Future of Innovation










