 The new electrocatalyst for hydrogen fuel cells consists of a thin platinum-cobalt alloy network and, unlike the catalysts commonly used today, does not require a carbon carrier.
The new electrocatalyst for hydrogen fuel cells consists of a thin platinum-cobalt alloy network and, unlike the catalysts commonly used today, does not require a carbon carrier.
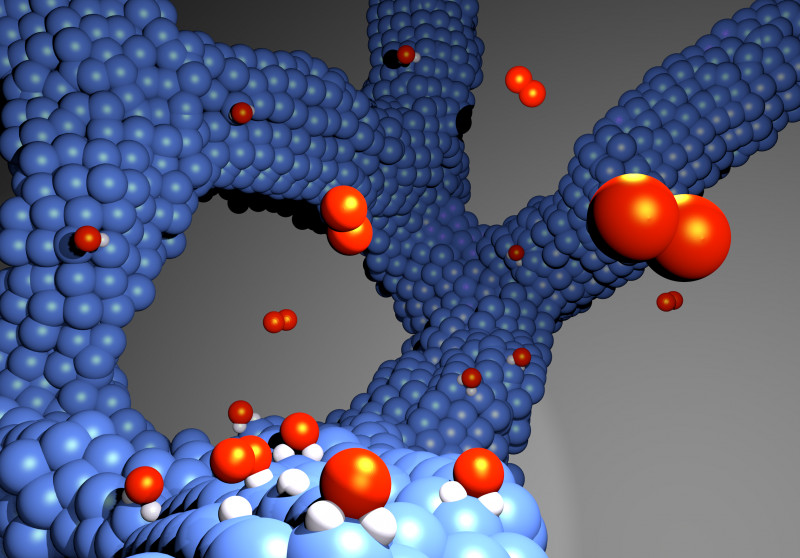
The new electrocatalyst for hydrogen fuel cells consists of a thin platinum-cobalt alloy network and, unlike the catalysts commonly used today, does not require a carbon carrier. Gustav Sievers - An international research team led by the University of Bern has succeeded in developing an electrocatalyst for hydrogen fuel cells which, in contrast to the catalysts commonly used today, does not require a carbon carrier and is therefore much more stable. The new process is industrially applicable and can be used to further optimize fuel cell powered vehicles without CO
2 emissions. Fuel cells are gaining in importance as an alternative to battery-operated electromobility in heavy traffic, especially since hydrogen is a CO
2-neutral energy carrier if it is obtained from renewable sources. For efficient operation, fuel cells need an electrocatalyst that improves the electrochemical reaction in which electricity is generated. The platinum-cobalt nanoparticle catalysts used as standard today have good catalytic properties and require only as little as necessary rare and expensive platinum. In order for the catalyst to be used in the fuel cell, it must have a surface with very small platinum-cobalt particles in the nanometer range, which is applied to a conductive carbon carrier material.
TO READ THIS ARTICLE, CREATE YOUR ACCOUNT
And extend your reading, free of charge and with no commitment.
Your Benefits
- Access to all content
- Receive newsmails for news and jobs
- Post ads


