science wire
Pharmacology
Results 3651 - 3700 of 4243.
Pharmacology - Health - 20.11.2015
Novartis Pharmaceuticals Corporation finalizes settlement agreement resolving civil suit filed by the U.S. Attorney’s Office for the Southern District of New York
Novartis Pharmaceuticals Corporation finalizes settlement agreement resolving civil suit filed by the U.S. Attorney's Office for the Southern District of New York Novartis Pharmaceutica
Pharmacology - Health - 18.11.2015
Regulatory submission for Sandoz’ proposed biosimilar pegfilgrastim accepted by the FDA
Regulatory submission for Sandoz' proposed biosimilar pegfilgrastim accepted by the FDA Sandoz demonstrates commitment to oncology by seeking approval for its proposed biosimilar pegfilgrastim.
Pharmacology - Health - 17.11.2015
Superiority of Novartis’ Ultibro Breezhaler over Seretide in reducing COPD exacerbations
Ultibro Breezhaler met primary endpoint and demonstrated superiority to Seretide in reducing COPD exacerbations during 52 weeks of treatment First large-scale study to confirm Ultibro Breezhaler is an effective steroid-free option that both reduces exacerbations and improves lung function in COPD patients with one or more exacerbations in the past year, compared to Seretide Full FLAME study results to be shared in 2016 Basel, November 17, 20
Health - Pharmacology - 15.11.2015
Alcon introduces Contoura Vision as first personalized LASIK procedure at American Academy of Ophthalmology Annual Meeting
Nearly 65% of eyes treated with personalized topography-guided LASIK experienced 20/16 vision or better in clinical study Alcon is the first refractive laser manufacturer to receive FDA approval fo
Pharmacology - Health - 08.11.2015
New two year data for Novartis' Cosentyx show sustained response and no progression in spinal damage as shown by x-ray in up to 80% of patients with ankylosing spondylitis
New two year data for Novartis' Cosentyx show sustained response and no progression in spinal damage as shown by x-ray in up to 80% of patients with ankylosing spondylitis A sub-study showed up to 80% of ankylosing spondylitis (AS) patients treated with Cosentyx had no radiographic progression in the spine over two years Cosentyx demonstrated a sustained response for signs and symptoms of AS in anti-TNF naïve patients with around 80%* achievin
Pharmacology - Health - 08.11.2015
Novartis presents new two year data for Cosentyx showing no progression in joint damage in 84% of psoriatic arthritis patients
New x-ray assessment data, from a sub-study, showed no progression of joint structural damage in the majority of patients with psoriatic arthritis over two years Psoriatic arthritis patients on Cosentyx maintained treatment response in joint and skin disease, physical function and quality of life over two years Cosentyx is the first interleukin-17A inhibitor to demonstrate efficacy in Phase III studies of patients with psoriatic arthritis , The
Pharmacology - Economics - 27.10.2015
Novartis delivered strong core margin expansion (cc) and continued to strengthen the pipeline in Q3; on track for full-year guidance
Solid growth (cc ) in Q3 sales, core operating income, core EPS for continuing operations Net sales were USD 12.3 billion (-6%, +6% cc) Operating income was USD 2.2 billion (-18%, +2% c
Pharmacology - Health - 23.10.2015
Novartis receives positive CHMP opinion for the first IL-17A inhibitor Cosentyx(TM) to treat ankylosing spondylitis and psoriatic arthritis
Novartis receives positive CHMP opinion for the first IL-17A inhibitor Cosentyx(TM) to treat ankylosing spondylitis and psoriatic arthritis Cosentyx (secukinumab) is recommended for approval in Europe for the treatment of ankylosing spondylitis (AS) and psoriatic arthritis (PsA) patients Cosentyx demonstrated rapid onset of action and long-term sustainability in patients with and without prior treatment using anti-tumor-necrosis-factor (anti-T
Pharmacology - Health - 21.10.2015
Novartis broadens immuno-oncology pipeline with acquisition of Admune Therapeutics and licensing agreements with XOMA and Palobiofarma
With four programs currently in clinical trials and five more expected to enter the clinic by the end of 2016, Novartis has rapidly built a robust portfolio of programs focused on stimulating the body's immune system to combat cancers.
Health - Pharmacology - 21.10.2015
Heart catheterization: Change in methods lowers mortality
The groin is the usual access point for investigating or treating the heart with a catheter, but using the wrist as access point reduces bleeding and lowers mortality. These findings are from an international study with major involvement from the University of Bern published today in the Lancet. About 46,000 cardiac catheterizations are carried out each year in Switzerland; about 22,000 of involve treatments of coronary vessels.
Health - Pharmacology - 15.10.2015
Kenya first country to launch 'Novartis Access', expanding affordable treatment options against chronic diseases
Novartis Access is a first-of-its-kind portfolio of products aimed at increasing access to medicines against cardiovascular diseases, diabetes, respiratory illnesses and breast cancer in lowand low-m
Pharmacology - Health - 10.10.2015
Novartis presents new data showing that the majority of patients are able to maintain clear or almost clear skin with Cosentyx across 3 years
Novartis presents new data showing that the majority of patients are able to maintain clear or almost clear skin with Cosentyx across 3 years Late-breaking data at EADV show that 8 out of 10 of psori
Pharmacology - Health - 08.10.2015
Long-term efficacy of Gilenya reinforced by new 'no evidence of disease activity' (NEDA-4) analysis in MS patients over seven years
Long-term efficacy of Gilenya reinforced by new 'no evidence of disease activity' (NEDA-4) analysis in MS patients over seven years NEDA-4 - no evidence of disease activity based on four parameters - relapses, MRI lesions, brain shrinkage and disability progression - is a comprehensive measure of MS disease control 31.2% to 44.8% of patients continuously treated with Gilenya in the FREEDOMS core and extension trials achieved NEDA
Health - Pharmacology - 05.10.2015
Alcon receives FDA Approval for pre-loaded intraocular lens delivery system to treat patients undergoing cataract surgery
AcrySof IQ Aspheric Intraocular Lens (IOL) with UltraSert TM Pre-loaded Delivery System helps maintain integrity of incision while facilitating smooth IOL delivery Follows earlier FDA approval of AcrySof IQ ReSTOR +2.
Pharmacology - Health - 02.10.2015
FDA accepts Sandoz regulatory submission for a proposed biosimilar etanercept
Etanercept is an anti-TNF medicine used to treat a range of immunological diseases including rheumatoid arthritis and psoriasis.
Health - Pharmacology - 01.10.2015
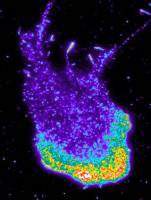
When cells from the connective tissue collide, they repel one another - this phenomenon was discovered more than 50 years ago. It is only now, however, that researchers at the University of Basel have discovered the molecular basis for this process, as they report in the journal Developmental Cell. Their findings could have important implications for cancer research.
Pharmacology - Health - 01.10.2015
Novartis announces NEJM publication of secukinumab Phase III data confirming significant efficacy in patients with psoriatic arthritis
In the FUTURE 1 study, secukinumab showed rapid and significant efficacy in active psoriatic arthritis (PsA) patients, including improvement of skin and joint disease and reduction in progression of joint structural damage Clinical benefits with secukinumab observed in patients with previous treatment with standard of care anti-TNF therapy and those without prior treatment Secukinumab is the first IL-17A inhibitor to report positive Phase III re
Health - Pharmacology - 28.09.2015
Patients with aggressive form of melanoma lived for more than two years on average when taking Novartis therapies Tafinlar + Mekinist
Phase III data showed median overall survival of 25.6 months in patients with BRAF+ V600E/K metastatic melanoma who received Tafinlar + Mekinist Tafinlar + Mekinist combination also demonstrat
Health - Pharmacology - 27.09.2015
Novartis drug Afinitor significantly improves progression-free survival in advanced nonfunctional gastrointestinal and lung NET
In pivotal study, everolimus reduced risk of disease progression by 52%; showed 11.0-month median progression-free survival vs 3.9 months for placebo Advanced, progressive, nonfunctional neuroendocrine tumors of GI or lung origin are rare forms of cancer with poor prognoses and limited treatment options ,[3, RADIANT-4 results highlighted at key European cancer congress; worldwide regulatory filings are underway Basel, September 27,
Health - Pharmacology - 25.09.2015
Novartis' new heart failure medicine Entresto(TM) recommended by CHMP for EU approval
Positive opinion from EU review body puts Entresto on track to be approved for HFrEF patients across Europe likely by year end Entresto was studied in world's largest heart failure trial which was stopped early on strength of results that showed a 20% cut in cardiovascular deaths vs enalapril Every day 10,000 Europeans are diagnosed with heart failure and 15 million already live with the condition, facing a high risk of death and poor quality of
Health - Pharmacology - 24.09.2015
Novartis appoints James E. Bradner, MD as President of the Novartis Institutes for BioMedical Research as Mark Fishman retires
New leader to continue to drive long-term innovation in Novartis research in wide range of therapeutic areas Basel, September 24, 2015 - Novartis announced today that Dr. James (Jay) E.
Pharmacology - Health - 24.09.2015
Antidepressants plus blood-thinners slow down brain cancer
24. EPFL scientists have found that combining antidepressants with anticoagulants slows down brain tumors (gliomas) in mice. Gliomas are aggressive brain tumors arising from the brain's supporting glial cells. They account for about a third of all brain tumors, and hold the highest incidence and mortality rate among primary brain cancer patients, creating an urgent need for effective treatments.
Health - Pharmacology - 18.09.2015
Swissmedic approves Novartis' new heart failure medicine Entresto(TM)
Decision by Swiss Health Authority follows US Food and Drug Administration (FDA) approval of Entresto in July, serves as an important reference approval for multiple other health authorities Entresto is indicated in Switzerland to reduce the risk of cardiovascular mortality and morbidity in patients with heart failure with reduced ejection fraction People with heart failure face high risk of death and poor quality of life, with only half al
Life Sciences - Pharmacology - 17.09.2015
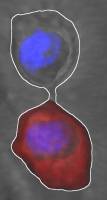
Neural stem cells generate new neurons throughout life in the mammalian brain. However, with advancing age the potential for regeneration in the brain dramatically declines. Scientists of the University of Zurich now identified a novel mechanism of how neural stem cells stay relatively free of aging-induced damage.
Life Sciences - Pharmacology - 08.09.2015
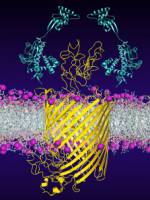
During their formation within the cells, many proteins rely on the assistance of protectors, so-called chaperones. They help the proteins to fold correctly and thus ensure the right final structure. The roles of chaperones in membrane protein folding have long remained unclear. Researchers at the Biozentrum, University of Basel, and at ETH Zurich now show how chaperones stabilize an immature bacterial membrane protein and guide it in the right folding direction, thus protecting it from misfolding.
Life Sciences - Pharmacology - 07.09.2015
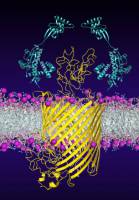
During their formation within the cells, many proteins rely on the assistance of molecular protectors, so-called chaperones. They help the proteins to fold correctly and thus ensure the right final structure. The roles of chaperones in membrane protein folding have long remained unclear. Researchers at the Biozentrum, University of Basel, and at ETH Zurich have now shown how chaperones stabilize an immature bacterial membrane protein and guide it in the right folding direction, thus protecting it from misfolding.
Health - Pharmacology - 04.09.2015
Novartis receives EU approval for Farydak , the first in its class of anticancer agents approved for patients with multiple myeloma
Farydak (panobinostat) combination is approved in the EU for patients with multiple myeloma who received >= 2 prior regimens including bortezomib and IMiD In clinical trials, Farydak combination i
Pharmacology - Health - 03.09.2015
Sandoz launches ZarxioTM (filgrastim-sndz), the first biosimilar in the United States
Launch follows March 6, 2015 FDA approval Sandoz One Source TM offers patient support services The digital press release with multimedia content can be accessed here: Holzkirchen, September 3, 2015 - Sandoz, a Novartis company, announced today that Zarxio(TM) (filgrastim-sndz) is now available in the United States.
Health - Pharmacology - 02.09.2015
Novartis receives EU approval for new Revolade use as first-in-class therapy for patients with severe aplastic anemia
Revolade is the first approved therapy in the EU for patients with severe aplastic anemia (SAA) who have not responded to other treatments Patients living with SAA, a rare blood disorder, have limi
Pharmacology - Physics - 02.09.2015
Preventing crystallization to improve drug efficiency
02. Esther Amstad and an international team of researchers have developed a method to increase the solubility of poorly soluble substances, such as many of the newly developed drugs. "Each year, pharmaceutical companies refuse many drugs during early stages of their development because of their poor solubility in the body which prevents their efficient uptake by the human body" explains Esther Amstad.
Pharmacology - Health - 01.09.2015
Novartis announces global partnership with Amgen to develop and commercialize pioneering neuroscience treatments
The companies plan to co-develop and co-commercialize a BACE inhibitor program in Alzheimer's Disease (AD); Novartis' oral therapy CNP520 will be the lead molecule Novartis and Amgen also plan to c
Health - Pharmacology - 01.09.2015
Novartis receives EU approval for Tafinlar and Mekinist , first combination approved for patients with aggressive form of melanoma
Approval based on two Phase III studies demonstrating statistically significant overall survival benefit with combination therapy vs BRAF inhibitor monotherapy Targeted combination offers BRAF V600
Health - Pharmacology - 28.08.2015
Novartis continues commitment to go the last mile in effort to eliminate leprosy
New five-year commitment includes donation of treatments worth more than USD 40 million and is expected to reach an estimated 1.3 million patients Novartis Foundation's new leprosy tracing pro
Economics - Pharmacology - 27.08.2015
Novartis International BioCamp highlights how collaboration and diversity drive innovation and inspires future science leaders
Health - Pharmacology - 24.08.2015
FDA expands use of Novartis drug Promacta to include treatment of children ages 1 and older with chronic immune thrombocytopenia
New oral suspension formulation, designed for younger children with rare blood disorder, is now approved For about one in four children with ITP, the condition persists for more than 12 months afte
Pharmacology - Health - 21.08.2015
Novartis acquires all remaining rights to GSK's Ofatumumab to develop treatments for MS and other autoimmune indications
Novartis strengthens multiple sclerosis focus with the addition of Ofatumumab to leading MS portfolio which includes Gilenya and investigational treatments BAF312 and CJM112 Ofatumumab is a fully hum
Pharmacology - Health - 20.08.2015
Novartis drug Odomzo gains EU approval for locally advanced basal cell carcinoma, providing new non-invasive therapy for patients
Approval follows positive CHMP opinion based on pivotal Phase II study showing durable objective response rate per central review of 56% in patients with laBCC Basal cell carcinoma is the most comm
Pharmacology - Health - 17.08.2015
Pivotal Phase II study showed Roche’s investigational immunotherapy atezolizumab shrank tumours in people with a specific type of lung cancer
Pivotal Phase II study showed Roche's investigational immunotherapy atezolizumab shrank tumours in people with a specific type of lung cancer Roche will discuss results with the U.S. Food and Drug Administration (FDA) as part of atezolizumab's Breakthrough Therapy Designation in lung cancer Roche today announced that in the large pivotal Phase II study, BIRCH, the investigational cancer immunotherapy atezolizumab (MPDL3280A; anti-P
Economics - Pharmacology - 03.08.2015
Novartis completes divestiture of influenza vaccines business to CSL Limited for USD 275 million
Pharmacology - Health - 29.07.2015
Novartis partners with Phase 4 Partners and institutional investors to help create Mereo BioPharma Group Ltd
Novartis is spinning off three mid stage clinical assets to Mereo for further development in exchange for equity Novartis will have a stake in the success of the development of these compounds, inclu
Health - Pharmacology - 28.07.2015
Stress Hormone Reduces Heroin Cravings
Every addiction is characterized by a strong desire for a certain addictive substance, be it nicotine, alcohol or other drug. Researchers at the University of Basel recently conducted a study on heroin addiction and demonstrated that the stress hormone cortisol can reduce addictive cravings. The findings from the research have been published in the medical journal Translational Psychiatry.
Pharmacology - Health - 24.07.2015
FDA approves Novartis drug Odomzo (sonidegib) for locally advanced basal cell carcinoma (laBCC), a form of skin cancer
Approval is based on pivotal Phase II study in which objective response rate (ORR) in patients with laBCC was 58%; responses were durable Basal cell carcinoma, the most common form of skin cancer,
Health - Pharmacology - 24.07.2015
Novartis drug Revolade recommended by CHMP for EU approval to treat patients with severe aplastic anemia, a serious blood disorder
If approved, Revolade would be the first treatment option in its class in the EU for certain patients with SAA Approximately 40% of SAA patients unresponsive to initial immunosuppressive therapy (I
Health - Pharmacology - 24.07.2015
Novartis combination therapy Tafinlar and Mekinist achieves important EU and US regulatory milestones
CHMP positive opinion for EU approval of Tafinlar and Mekinist combination in BRAF V600 mutation-positive melanoma, the most aggressive type of skin cancer FDA grants priority review for full appro
Pharmacology - Economics - 21.07.2015
Novartis delivered solid performance in the second quarter, with strong innovation and progress on new launches
Sales , core operating income and core EPS grew (cc ) for continuing operations in Q2: Net sales amounted to USD 12.7 billion (-5%, +6% cc) Operating income was USD 2.3 billion (-28%,
Pharmacology - Health - 16.07.2015
Novartis malaria treatment Coartem 80/480mg receives WHO prequalification, enabling greater access for patients
WHO prequalified Coartem 80/480mg is the first and only high strength malaria treatment available for donor-funded public sector procurement Coartem 80/480mg reduces the pill burden for patients,
Health - Pharmacology - 13.07.2015
In a pivotal study, Roche’s investigational immunotherapy atezolizumab shrank tumours in people with a specific type of bladder cancer
In a pivotal study, Roche's investigational immunotherapy atezolizumab shrank tumours in people with a specific type of bladder cancer Results showed that high levels of PD-L1 expression were associated with greater responses to atezolizumab Roche will discuss results with the U.S. Food and Drug Administration (FDA) as part of atezolizumab's Breakthrough Therapy Designation in bladder cancer Roche today announced that in the IMvigo
Pharmacology - Health - 07.07.2015
Novartis' new heart failure medicine LCZ696, now called Entresto(TM), approved by FDA to reduce risk of cardiovascular death and heart failure hospitalization
Novartis' new heart failure medicine LCZ696, now called Entresto(TM), approved by FDA to reduce risk of cardiovascular death and heart failure hospitalization First in the world approval brings hope
Pharmacology - Innovation - 30.06.2015
Novartis Pharmaceuticals launches the first app for visually impaired people for use with the Apple Watch and other smart watches
New features of the ViaOpta apps, previously available for iPhone and Android TM mobile phones, are also available to enhance the daily lives of people with visual impairments These apps foster ind
Pharmacology - Health - 29.06.2015
Novartis announces publication in The Lancet showing sustained efficacy with secukinumab over one year in psoriatic arthritis patients
In the FUTURE 2 study, secukinumab demonstrated rapid onset of action, was significantly superior to placebo in improving signs and symptoms of psoriatic arthritis (PsA), with efficacy sustained over one year Secukinumab is the first IL-17A inhibitor to significantly improve joint and skin symptoms of PsA, and provides physical functioning and quality of life benefits Global regulatory submissions have been filed for secukinumab in PsA and ank
Life Sciences - Mar 27
Understanding the Brain - TU Ilmenau's EU EMBRACE Project Nominated for European Excellence Award
Understanding the Brain - TU Ilmenau's EU EMBRACE Project Nominated for European Excellence Award
Social Sciences - Mar 27
A manual addresses, for the first time in Spain, child and adolescent sexual exploitation
A manual addresses, for the first time in Spain, child and adolescent sexual exploitation

Environment - Mar 26
Changing vegetation in thawing permafrost increases emissions of greenhouse gases
Changing vegetation in thawing permafrost increases emissions of greenhouse gases

Environment - Mar 26
University of Manchester hits major sustainability milestone, with Main Campus becoming 100% 'Zero Landfill'
University of Manchester hits major sustainability milestone, with Main Campus becoming 100% 'Zero Landfill'

Social Sciences - Mar 26
"It would be naive to believe that a social media ban will solve all problems"
"It would be naive to believe that a social media ban will solve all problems"

Health - Mar 26
Earlier detection, better outcomes: Irish researchers target rising bowel cancer rates with new blood test
Earlier detection, better outcomes: Irish researchers target rising bowel cancer rates with new blood test
Environment - Mar 26
UK must improve energy efficiency to end 50 years of policy failure and prevent future energy crises, study argues
UK must improve energy efficiency to end 50 years of policy failure and prevent future energy crises, study argues

Mathematics - Mar 26
From Materials to Medical Imaging, Fonseca's Work Shapes the Future of Innovation
From Materials to Medical Imaging, Fonseca's Work Shapes the Future of Innovation










