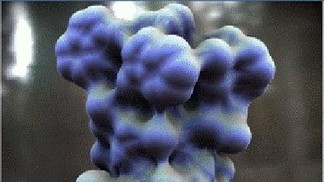
 Part of the gramicidin molecule
Part of the gramicidin molecule
EPFL chemists have shown how the three-dimensional shape of an antibiotic changes when it is in an aqueous environment. This could lead to a better understanding of how drugs interact with biological molecules. Like a key inserted into a lock, the molecules in drugs bind with and act upon biomolecules. The more precisely we know these molecules' three-dimensional structure, the better we will understand how their active components work. Biomolecules often exist in aqueous environments; this is the case in the human body. But water isn't just a neutral environment; it can also interact with the molecules and change their structure. Scientists from EPFL's Laboratory of Molecular Physical Chemistry have shown how the 3D structure of gramicidin - a natural antibiotic - changes depending on the number of water molecules surrounding it.
TO READ THIS ARTICLE, CREATE YOUR ACCOUNT
And extend your reading, free of charge and with no commitment.
Your Benefits
- Access to all content
- Receive newsmails for news and jobs
- Post ads


