 Prof. Armido Studer with Dr. Hui Cao and Dr. Qiang Cheng (from left). The flipchart shows the chemical transformation of loratadine, an important histamine H1-receptor antagonist modified through a trifluoromethyl group.
Prof. Armido Studer with Dr. Hui Cao and Dr. Qiang Cheng (from left). The flipchart shows the chemical transformation of loratadine, an important histamine H1-receptor antagonist modified through a trifluoromethyl group.
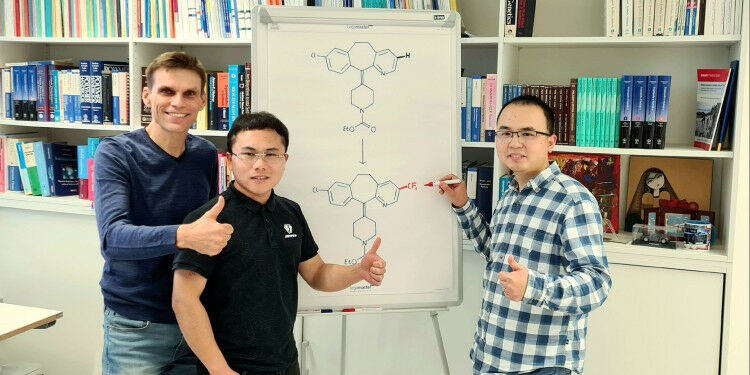
Prof. Armido Studer with Dr. Hui Cao and Dr. Qiang Cheng ( from left ). The flipchart shows the chemical transformation of loratadine, an important histamine H1-receptor antagonist modified through a trifluoromethyl group. Münster University - Studer working group - In chemicals used in agriculture, as well as in pharmaceuticals and a variety of materials, pyridines are often found as so-called functional units which decisively determine the chemical properties of substances. Pyridines belong to the group of ring-shaped carbon-hydrogen (C'H) compounds ("heterocycles"), and they contain a nitrogen atom (N). For chemists, the direct functionalization of the carbon-hydrogen bonds (C'H bonds) of pyridines is a straightforward approach to designing and modifying complex molecules, including in the final stage of the synthesis sequence. This latter means that active ingredients can be chemically modified without having to build them up anew. The functionalization of the pyridine in a certain position in relation to the nitrogen atom - in the difficult-to-access "meta-position" - is extremely challenging and rare.
TO READ THIS ARTICLE, CREATE YOUR ACCOUNT
And extend your reading, free of charge and with no commitment.
Your Benefits
- Access to all content
- Receive newsmails for news and jobs
- Post ads


