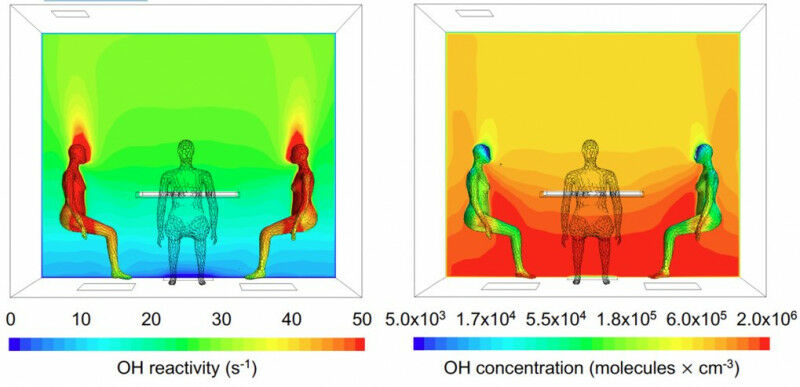
 Computer modelling of the OH reactivity (left) and OH concentration (right) around human bodies in a typical indoor situation while people sitting around a table.
Computer modelling of the OH reactivity (left) and OH concentration (right) around human bodies in a typical indoor situation while people sitting around a table.
People generate their own oxidation field and change the indoor air chemistry around them. Computer modelling of the OH reactivity ( left ) and OH concentration ( right ) around human bodies in a typical indoor situation while people sitting around a table. UC Irvine - People typically spend 90 percent of their lives inside, at home, at work or in transport. Within these enclosed spaces, occupants are exposed to a multitude of chemicals from various sources, including outdoor pollutants penetrating indoors, gaseous emissions from building materials and furnishings, and products of our own activities such as cooking and cleaning. In addition, we are potent mobile emission sources of chemicals that enter the indoor air from our breath and skin. But how do the chemicals disappear again? In the atmosphere outdoors, this happens to a certain extent naturally by itself, when it rains and through chemical oxidation. Hydroxyl (OH) radicals are largely responsible for this chemical cleaning.
TO READ THIS ARTICLE, CREATE YOUR ACCOUNT
And extend your reading, free of charge and with no commitment.
Your Benefits
- Access to all content
- Receive newsmails for news and jobs
- Post ads


