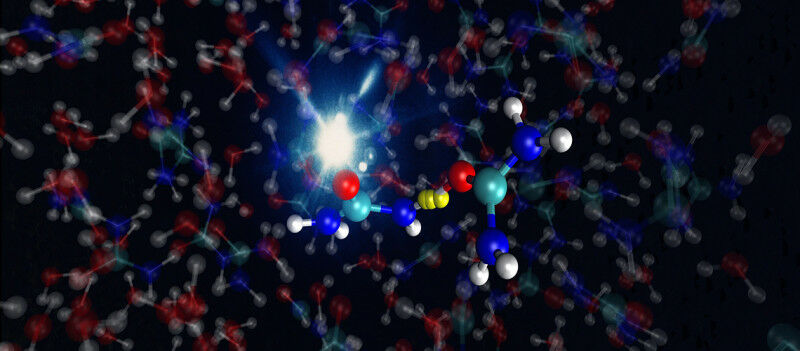
 Photoionization-induced proton transfer between two urea molecules in an aqueous urea solution..
Photoionization-induced proton transfer between two urea molecules in an aqueous urea solution..
Thanks to a new technique for observing chemical reactions in liquids, UNIGE and ETH are shedding light on one of the hypotheses about the chemical origins of life. Photoionization-induced proton transfer between two urea molecules in an aqueous urea solution. Ludger Inhester, DESY, Hamburg What processes led to the appearance of life on our planet? Some scientists believe that urea played a key role. After being enriched in warm puddles, this small molecule is thought to have been exposed to cosmic rays to produce malonic acid, the source of RNA and DNA. To observe how a urea solution exposed to such rays reacts, a team from the University of Geneva and ETH Zurich (ETH) developed a spectroscopic method for observing chemical reactions in liquids, with extremely high temporal resolution. These results, published in the journal Nature , shed new light on the appearance of life on Earth. The first evidence of life on Earth dates back 3.8 billion years.
TO READ THIS ARTICLE, CREATE YOUR ACCOUNT
And extend your reading, free of charge and with no commitment.
Your Benefits
- Access to all content
- Receive newsmails for news and jobs
- Post ads


