The study investigates epigenetic regulation by placental DNA methylation, opening new perspectives on the relationship between this process and disorders of pregnancy and embryonic development. They identify the crucial role in placental development of a protein, which together with other enzymes, restores partially methylated domains in human trophoblast stem cells.
Gene expression is the process by which the information contained in genetic material is used to produce proteins, which in turn perform a function in the cell. The regulation of gene expression allows cells to specialize and respond to changes in their environment, processes that are fundamental to embryonic development and the functioning of organisms. Among the mechanisms that control gene expression is DNA methylation, an epigenetic process that affects which genes are turned on or off, without changing their genetic sequence.
In this context, the team of Dr. Vicente Pérez García, from the Centro de Biología Molecular Severo Ochoa (CBM-CSIC-UAM), in collaboration with Dr. Courtney Hanna, from the Loke Centre for Trophoblast Research at the University of Cambridge, have deciphered a key mechanism related to DNA methylation in the placenta, published in the prestigious journal Cell Stem Cell.
A unique and relevant methylome
The placental methylome, or set of placental DNA methylation patterns, is an epigenetic signature unique to the placenta, characterized by the presence of partially methylated domains. This pattern is highly conserved among mammals and shares similarities with methylomes observed in many types of cancer. In addition, it has been closely linked to pregnancy complications, making it a focus of great scientific and medical interest.
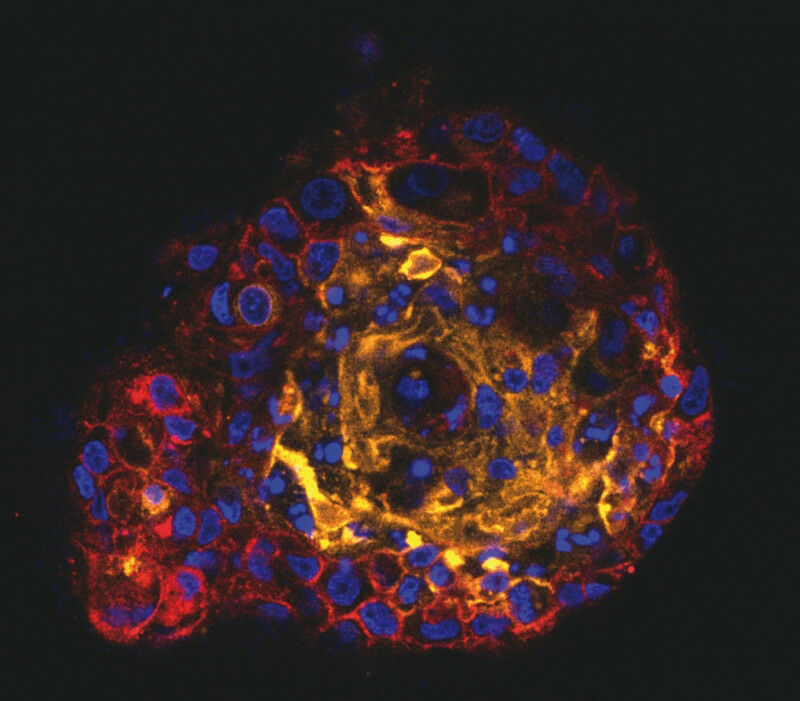
"To explore these epigenetic characteristics, we used human trophoblast stem cells, which are an experimental model derived from the placenta during its early development," says Vicente Perez, corresponding author of the study. "These cells have the capacity to differentiate into the different cell types that form the placenta. Trophoblast stem cells allow us to study placental development in the laboratory."
However, the study revealed that these cells do not accurately reproduce the methylome of the early human primary trophoblast, as they lack the partially methylated domains characteristic of this tissue.
The crucial role of DNMT3L
The most relevant finding of the study was the absence of the DNMT3L protein, which enhances the catalytic activity of the DNA-methylating enzymes DNMT3A and DNMT3B in trophoblast stem cells. This enzyme is essential for establishing the partially methylated domains in the mouse trophoblast and apparently also plays a conserved role in humans. To test this, the researchers introduced ectopic expression of DNMT3L into trophoblast stem cells, which restored the partially methylated domains characteristic of the placental methylome. This finding confirms the importance of DNMT3L in de novo methylation during trophoblast development and suggests a central role in the epigenetic regulation of placental development.
Implications and future
This advance opens new perspectives for understanding the role of partially methylated domains in placental development and their relationship to pregnancy complications. It also underscores the value of trophoblast stem cells as an experimental model and the need for further refinement of these tools to better reflect the epigenetic conditions of the early trophoblast.
The study represents a significant step toward understanding the epigenetic mechanisms underlying human development and their implications for reproductive health.
Bibliographic reference:
Georgia Lea, Paula Doria-Borrell, Ana Ferrero-Micó, Anakha Varma, Claire Simon, Holly Anderson, Laura Biggins, Katrien De Clercq, Simon Andrews, Kathy K. Niakan, Lenka Gahurova, Naomi McGovern, Vicente Pérez-García*, Courtney W. Hanna*. Ectopic expression of DNMT3L in human trophoblast stem cells restores features of the placental methylome, Cell Stem Cell, 2025, DOI: https://doi.org/10.1016/j.stem.2024.12.007
More scientific culture in UAM Gazette



