Scientists from the Institute of Oncology Research and the Institute for Research in Biomedicine, both affiliated with USI, report in Cell Reports the discovery of the first drugs that inhibit FOXA1 and FOXA2 - two master gene regulators that control cancer cell identity and growth in prostate cancer. The study reveals a new therapeutic vulnerability in both early and advanced forms of the disease.
Master regulators of prostate cancer progression
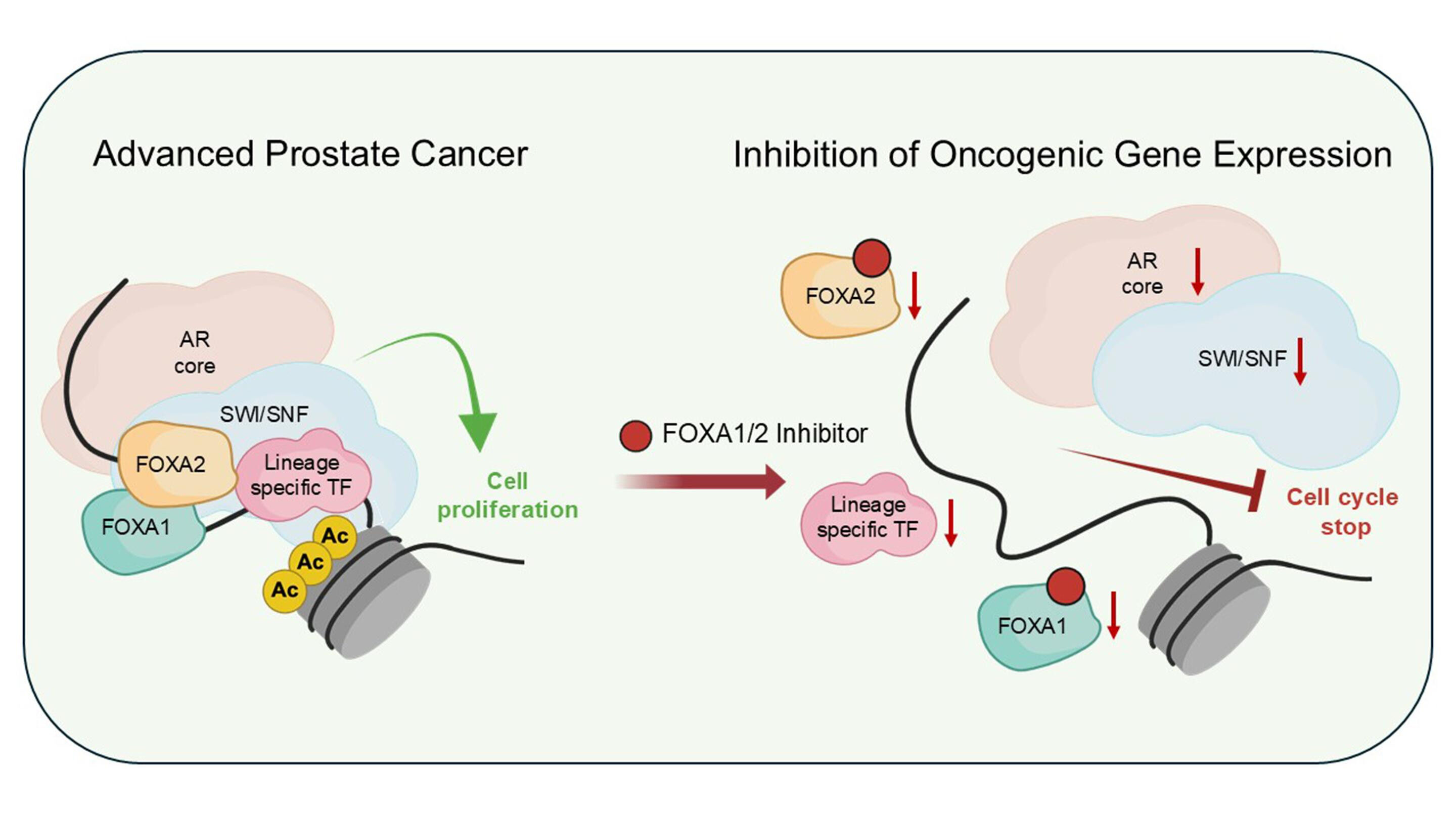
Prostate cancer growth is primarily driven by the androgen receptor (AR), a signaling pathway that promotes tumor development and is the main therapeutic target today. The transcription factors FOXA1 and FOXA2 act as “gene switches” that control how cancer cells respond to AR signaling. While FOXA1 supports the AR-dependent form of the disease, FOXA2 takes over in more advanced, treatment-resistant cancers that no longer rely on AR activity.
Blocking both factors halts cancer cell growth
The research team discovered that FOXA1 and FOXA2 can cooperate to sustain cancer cell proliferation even without AR signaling. When both are inhibited—either through genetic suppression or new chemical inhibitors - cancer cells lose their lineage identity and stop dividing. This finding identifies a shared molecular weakness across different prostate cancer subtypes.
Expanding treatment options for advanced prostate cancer
The development of these first-in-class inhibitors marks a breakthrough in targeting transcription factors, which were long considered “undruggable.” By blocking FOXA1 and FOXA2, researchers open the door to therapies that could benefit patients with both hormone-sensitive and resistant prostate cancers. The study establishes a new conceptual framework for treating lineage-plastic cancers through disruption of their core gene regulatory networks.
Reference:
Targeting FOXA1 and FOXA2 disrupts the lineage-specific oncogenic output program in prostate cancer. Nicola Formaggio, Jacopo Sgrignani, Gayathri Thillaiyampalam, Claudio Lorenzi, Giada A. Cassamagnago, Marco Coazzoli, Federico Costanzo, Yanick Uebelhart, Daniela Bossi, Diego Camuzi Cassiano, Andrea Rinaldi, Matteo Pecoraro, Roger Geiger, Raffaella Santoro, Marco Bolis, Andrea Cavalli, Jean-Philippe Theurillat. Published in Cell Reports.



