science wire
Pharmacology
Results 3601 - 3650 of 4243.
Health - Pharmacology - 19.05.2016
Novartis announces investment in FortiHFy clinical program of Entresto and heart failure
Forti fying H eart F ailure clinical evidence and patient qualit y of life (FortiHFy) is an umbrella clinical program comprising over 40 active or planned trials The global clinical program will generate additional data on symptom reduction, efficacy, safety, quality of life benefits and real world evidence with Entresto Investigators and patients from over 50 countries to participate over 5 years - Novartis today announced the establishment
Pharmacology - Health - 19.05.2016
Phase III study of Roche’s Alecensa (alectinib) showed superior efficacy versus crizotinib in Japanese people with a specific type of lung cancer
Phase III study of Roche's Alecensa (alectinib) showed superior efficacy versus crizotinib in Japanese people with a specific type of lung cancer First investigational head-to-head study of Alecensa versus crizotinib in people with advanced ALK-positive non-small cell lung cancer (NSCLC) Roche today announced that Alecensa , an oral anaplastic lymphoma kinase (ALK) inhibitor, reduced the risk of disease worsening or death (progression free survi
Health - Pharmacology - 18.05.2016
MONALEESA-2 trial of Novartis’ LEE011 (ribociclib) stopped due to positive efficacy results at interim analysis in HR+/HER2- advanced breast cancer
MONALEESA-2 trial of Novartis' LEE011 (ribociclib) stopped due to positive efficacy results at interim analysis in HR+/HER2- advanced breast cancer Independent Data Monitoring Committee recommends st
Pharmacology - Health - 18.05.2016
Novartis to present pivotal data in hematologic and solid tumor cancers at 2016 ASCO Annual Meeting
First data from two treatment-free remission (TFR) studies of Ph+ CML patients treated with Tasigna both in front-line and second-line following Glivec * First genomic analysis and 3-year efficacy/
Pharmacology - Health - 17.05.2016
Novartis focuses Pharmaceuticals Division by creating two business units, Novartis Pharmaceuticals and Novartis Oncology; appoints leaders to the Executive Committee of Novartis
Health - Pharmacology - 15.05.2016
Major study published in NEJM confirms Novartis’ Ultibro Breezhaler superiority over Seretide in preventing COPD exacerbations
FLAME study showed consistent superiority of Ultibro Breezhaler over Seretide across exacerbation outcomes, lung function and health-related quality of life in COPD patients Ultibro Breezhaler also significantly reduced the rate and prolonged the time to the first moderate or severe exacerbation, compared to Seretide Results published in the New England Journal of Medicine are anticipated to have important implications for the care of peopl
Health - Pharmacology - 29.04.2016
Novartis drug Afinitor recommended by CHMP for European Union approval to treat select GI and lung neuroendocrine tumors
If approved, Afinitor would be first treatment in EU indicated for advanced progressive, nonfunctional lung NET and first oral therapy for this type of GI NET Advanced, progressive, nonfunctional GI
Health - Pharmacology - 28.04.2016

When medication is used to shut off the oxygen supply to tumor cells, the cells adapt their metabolism in the medium term - by switching over to producing energy without oxygen. This observation by biomedical scientists at the University of Basel and University Hospital Basel could be used for treatments that can inhibit tumor growth in the long term, as the researchers report in the latest issue of the journal Cell Reports.
Pharmacology - Health - 27.04.2016
Novartis receives three FDA Breakthrough Therapy Designations for Ilaris to treat rare types of Periodic Fever Syndromes
Subject to approvals, Ilaris will likely be first FDA-approved treatment for TRAPS and HIDS/MKD, and an important alternative treatment for patients with FMF Designations support potential expedite
Economics - Pharmacology - 21.04.2016
Novartis delivered solid Q1 despite Gleevec loss of exclusivity; investing behind new launches for long-term growth
Net sales up 1% (cc ), as Growth Products offset Gleevec impact Growth Products grew 24% (USD) to USD 3.9 billion, or 34% of Group net sales Cosentyx (USD 176 million) continues to grow strongl
Pharmacology - Health - 21.04.2016

Most cancer cells carry unique receptors on their surface. Because the receptors extend into the cell's interior, they act as intermediaries between the outside and the inside.
Health - Pharmacology - 20.04.2016
Novartis drug Afinitor significantly reduces seizures in Phase III study of patients with tuberous sclerosis complex
Everolimus is the first adjunctive therapy shown in a prospective randomized Phase III study to achieve clinically significant seizure control in TSC patients Seizures are the most common TSC-related neurological condition, yet about 60% of patients don't attain seizure control with available anti-epileptic therapies Results presented at AAN will be discussed with health authorities for potential worldwide regulatory filings - Novartis today
Health - Pharmacology - 12.04.2016
Roche to present new data about multiple sclerosis
Roche to present new data at AAN showing superior efficacy of investigational medicine ocrelizumab versus comparators on disease activity and progression in two forms of multiple sclerosis New analyses showing superior efficacy of ocrelizumab across clinical and subclinical outcomes compared with interferon beta- 1a (Rebif ) in people with relapsing MS and compared with placebo in primary progressive MS will be presented Roche announced today th
Health - Pharmacology - 12.04.2016
Roche announces FDA grants Venclexta(venetoclax) accelerated approval for people with hard-to-treat type of chronic lymphocytic leukemia
Roche announces FDA grants Venclexta(venetoclax) accelerated approval for people with hard-to-treat type of chronic lymphocytic leukemia Venclexta is designed to help restore a cell's ability to self-destruct and is the first medicine of its kind to be approved Roche announced today that the U.S. Food and Drug Administration (FDA) granted accelerated approval to Venclexta(TM) (venetoclax) for the treatment of people with chronic lymphocytic leukemia (CLL) with 17p deletion, as detected by an FDA approved test, who have received at least one prior therapy.
Health - Pharmacology - 11.04.2016
FDA grants priority review for Roche’s cancer immunotherapy atezolizumab in specific type of lung cancer
FDA grants priority review for Roche's cancer immunotherapy atezolizumab in specific type of lung cancer Roche today announced that the U.S. Food and Drug Administration (FDA) has accep
Pharmacology - Health - 08.04.2016
Sandoz receives EC approval for subcutaneous route of administration in biosimilar Binocrit’s nephrology indication
Pharmacology - Health - 07.04.2016
Novartis receives EU approval for Revolade as first-in-class therapy for children aged 1 year and above with chronic ITP
EU approval of Revolade expands treatment options for pediatric patients aged 1 year and above with chronic ITP who have not responded to other therapies Two formulations approved: once-daily table
Health - Pharmacology - 02.04.2016
New analyses show Novartis’ Entresto(TM) reduced cardiovascular death or hospitalization for heart failure, consistently benefitting patients with reduced ejection fraction regardless
New analyses show Novartis' Entresto(TM) reduced cardiovascular death or hospitalization for heart failure, consistently benefitting patients with reduced ejection fraction regardless.
Economics - Pharmacology - 31.03.2016
Novartis publishes updated 2015 segment financials reflecting new division structure
Updated segment financials reflect transfer of Ophthalmic Pharmaceuticals franchise from Alcon to Pharmaceuticals, and 19 mature products from Pharmaceuticals to Sandoz, as outlined in Q4 and FY 2015
Pharmacology - 31.03.2016
Roche to initiate testing for Zika virus at U.S. Blood Centres under FDA Investigational New Drug Application protocol
Roche to initiate testing for Zika virus at U.S. Blood Centres under FDA Investigational New Drug Application protocol New cobas Zika test will screen blood samples on the cobas 6800/8800 Systems Roche today announced that the U.S. Food and Drug Administration (FDA) has provided approval to initiate collection and testing of blood samples for screening with the cobas Zika assay under an Investigational New Drug Application (IND) protocol.
Pharmacology - Chemistry - 18.03.2016
Towards a mobile drugs lab
Pump out pills 24/7 and reduce wastage? Pharmaceutical companies are rolling out the red carpet for continuous manufacturing.
Life Sciences - Pharmacology - 15.03.2016

Natural products from bacteria and fungi are an important source of current and novel drugs, such as antibiotics.
Health - Pharmacology - 11.03.2016
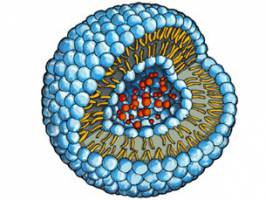
Liposomes are currently used as drug delivery vehicles but recognized by the immune system. Scientists from the universities of Basel and Fribourg have shown that special artificial liposomes do not elicit any reaction in human and porcine sera as well as pigs. The study was published in the Journal Nanomedicine: Nanotechnology, Biology, and Medicine.
Life Sciences - Pharmacology - 07.03.2016

Almost every second woman suffers from a bladder infection at some point in her life. Also men are affected by cystitis, though less frequently.
Pharmacology - Health - 05.03.2016
Novartis’ Cosentyx superior to Stelara in delivering long-lasting skin clearance (PASI 90) for psoriasis patients at 52 weeks
New data at AAD 2016 shows Cosentyx is significantly more efficacious than Stelara in sustaining skin clearance (PASI 90 to PASI 100) at 52 weeks Cosentyx delivered and sustained skin clearance (PASI 90 to PASI 100) in nearly 8 out of 10 moderate-to-severe psoriasis patients Cosentyx granted first-line systemic indication for psoriasis in Europe and recently approved in psoriatic arthritis and ankylosing spondylitis in the US and Europe The
Health - Pharmacology - 26.02.2016
FDA approves new indication for Novartis drug Afinitor for progressive, nonfunctional GI and lung neuroendocrine tumors (NET)
In advanced progressive, nonfunctional NET, Afinitor is the first approved treatment for patients with lung NET and the first oral therapy for GI NET Approval helps fulfill unmet need as progressiv
Pharmacology - Health - 19.02.2016
Novartis drug PKC412 (midostaurin) receives Breakthrough Therapy designation from the FDA for newly-diagnosed FLT3-mutated acute myeloid leukemia (AML)
Novartis drug PKC412 (midostaurin) receives Breakthrough Therapy designation from the FDA for newly-diagnosed FLT3-mutated acute myeloid leukemia (AML) PKC412 (midostaurin) significantly improved overall survival of adult patients eligible to receive standard induction and consolidation chemotherapy AML has the lowest survival rate of all adult leukemias and the treatment strategy with chemotherapy has remained unchanged for more than 25 years
Pharmacology - Health - 12.02.2016
Sandoz strengthens its biosimilars portfolio with acquisition of Pfizer’s biosimilar infliximab in EEA
Sandoz acquires rights to PF-06438179 (biosimilar infliximab) in the European Economic Area (EEA)* Sandoz plans to complete Phase III development program and file for registration in the EU Deal stre
Pharmacology - Health - 11.02.2016
Sandoz advances its biosimilars program with EMA acceptance of regulatory submission for biosimilar pegfilgrastim
Sandoz' late-stage biosimilar pipeline gains momentum with sustained investment - five of 10 planned regulatory filings achieved Sandoz believes that the totality of evidence in its submission, inclu
Pharmacology - Health - 04.02.2016

About thirty percent of all medical drugs such as beta-blockers or antidepressants interact with certain types of cell surface proteins called G protein coupled receptors. Researchers at the Biozentrum of the University of Basel, in collaboration with scientists from the Paul Scherrer Institute, have now elucidated in detail how the structure of such a receptor changes when drugs bind and how the structural change transmits a signal to the cellular interior.
Health - Pharmacology - 31.01.2016
Novartis Foundation commemorates World Leprosy Day with its continued efforts to go the last mile toward elimination
2016 marks 30 years of commitment by the Novartis Foundation to support people affected by leprosy and to end this ancient disease New strategy from the Novartis Foundation to accelerate elimination
Pharmacology - Economics - 27.01.2016
Novartis delivered strong sales growth and core margin expansion (cc[1]) in 2015; announces plans to accelerate growth at Alcon, streamline Group operations
Novartis delivered strong sales growth and core margin expansion (cc ) in 2015; announces plans to accelerate growth at Alcon, streamline Group operations Strong growth (cc) in full year sales, core o
Pharmacology - Health - 15.01.2016
Novartis receives two new FDA approvals for Cosentyx to treat patients with ankylosing spondylitis and psoriatic arthritis in the US
Cosentyx is the first and only interleukin-17A (IL-17A) inhibitor approved for adult patients with ankylosing spondylitis (AS) and psoriatic arthritis (PsA) FDA approval for Cosentyx is based on efficacy and safety outcomes shown across four Phase III studies, including over 1,500 patients with either AS or PsA In studies, Cosentyx met the primary endpoints showing statistically significant improvements versus placebo in the signs and sympto
Health - Pharmacology - 11.01.2016
Novartis continues to grow immuno-oncology pipeline through collaboration and licensing agreement with Surface Oncology
Agreement gives Novartis access to four pre-clinical programs that target regulatory'T cell populations, inhibitory cytokines, and immunosuppressive metabolites in the tumor microenvironment Novartis
Health - Pharmacology - 05.01.2016
Novartis Pharmaceuticals collaborates with Qualcomm in digital innovation with the Breezhaler(TM) inhaler device to treat COPD
The Breezhaler(TM) inhaler is the device for Novartis' portfolio of COPD treatments of Onbrez and Seebri, and Ultibro, the leading LABA/LAMA treatment The collaboration further supports Novartis' foc
Pharmacology - Health - 23.12.2015
Novartis announces NEJM publication of two major trials showing significant efficacy of Cosentyx in ankylosing spondylitis patients
Cosentyx (secukinumab) is the first IL-17A inhibitor approved in Europe to treat ankylosing spondylitis (AS) and first treatment advance in 16 years since anti-tumor necrosis factor (anti-TNF) therapy, the current standard of care Cosentyx provided improvements in the signs and symptoms, physical function and quality of life measures in active AS as early as Week 1 and through to Week 52 The study enrolled anti-TNF naïve patients and patients
Pharmacology - Health - 09.12.2015
300 million child-friendly antimalarial treatments supplied without profit by Novartis
Coartem Dispersible * , the first WHO prequalified pediatric antimalarial treatment, has become the standard of care in over 30 malaria-endemic countries Since 2009, Novartis has supplied 300 milli
Pharmacology - Health - 08.12.2015
Sandoz advances its biosimilars program with European Medicines Agency (EMA) acceptance of regulatory submission for biosimilar etanercept
Sandoz is seeking approval for all indications included in the reference product's label which includes chronic inflammatory conditions like rheumatoid arthritis and psoriasis.
Pharmacology - Health - 07.12.2015
Phase III data shows Sandoz’ proposed biosimilar pegfilgrastim has similar safety and efficacy as the reference product
PROTECT 2 study compared safety and efficacy of the proposed biosimilar pegfilgrastim with the reference product for the prevention of neutropenia in patients with breast cancer. The study met its primary endpoints demonstrating equivalence and non-inferiority to the reference product. Data presented at the 57th American Society of Hematology (ASH) Annual Meeting and Exposition.
Pharmacology - Health - 07.12.2015
New data from pivotal study showed Roche’s Gazyva/Gazyvaro induced deep remissions and provided meaningful quality of life improvements in people with difficult-to-treat indolent non-Hodgkin lymphoma
New data from pivotal study showed Roche's Gazyva/Gazyvaro induced deep remissions and provided meaningful quality of life improvements in people with difficult-to-treat indolent non-Hodgkin lymphoma Minimal residual disease-negativity rates were almost double for people receiving Gazyva/Gazyvaro plus bendamustine versus bendamustine alone in patients with follicular lymphoma Roche today announced follow-up results from the pivotal phase III GAD
Pharmacology - Health - 07.12.2015
Novartis highlights new CTL019 Phase II data demonstrating 93% complete remission in pediatric patients with r/r ALL
55 of 59 patients experienced complete remissions in a single-arm study of the largest investigational CART therapy in children and young adults Additional data presented include analyses on safety and efficacy of CTL019 in r/r ALL and other CART therapies in r/r ALL and AML , , , - The latest findings from an ongoing Phase II study of CTL019, an investigational chimeric antigen receptor'T cell (CART) therapy, further support its potential in the treatment of children and young adults with relapsed/refractory acute lymphoblastic leukemia (r/r ALL).
Pharmacology - Health - 06.12.2015
Novartis drug PKC412 (midostaurin) improves overall survival by 23% in global Phase III study of AML patients with FLT3 mutations
Study in partnership with the Alliance for Clinical Trials in Oncology is the first large controlled trial to show overall survival benefit in FLT3-mutated AML AML is the most common acute leukemia
Pharmacology - Health - 06.12.2015
Novartis announces new CTL019 study data demonstrating overall response in adult patients with certain types of lymphoma
Overall response rate at 3 months was 47% (7/15) in diffuse large B-cell lymphoma and 73% (8/11) in follicular lymphoma Successful technology transfer enabling commercial scale-up of CTL019 cell processing to support global trials reported in other ASH presentation Novartis global Phase II CTL019 clinical trials expanded to EU, Canada and Australia, marking first industry-sponsored CART trials to be opened outside US - Findings from an ongoin
Health - Pharmacology - 05.12.2015
Novartis announces Phase III studies of Jakavi show disease improvement in patients with myelofibrosis and polycythemia vera
Data suggest myelofibrosis (MF) patients treated with Jakavi (ruxolitinib) after 5 years lived longer despite crossover from best available therapy after week 48 Phase III results show Jakavi met p
Pharmacology - Health - 02.12.2015
Novartis presents new data on targeted combination therapy at SABCS reinforcing commitment to breast cancer patients
More than 50 abstracts will be presented, including data on Afinitor and investigational compounds for advanced breast cancer, showing pipeline strength Presentations highlight results of combinati
Pharmacology - Health - 30.11.2015
Novartis highlights clinical advances at ASH 2015, underscoring leadership in hematology research
Pivotal overall survival data for investigational compound PKC412 (midostaurin) in adults with newly diagnosed FLT3-mutated acute myeloid leukemia Presentations highlight new CTL019 data in relapsed/
Health - Pharmacology - 26.11.2015
Antibody-Drug Compounds and Immunotherapy to Treat Breast Cancer
To more efficiently treat breast cancer, scientists have been researching molecules that selectively bind to cancer cells and deliver a substance that can kill the tumor cells, for several years. Researchers from the University and University Hospital Basel have now for the first time successfully combined such an antibody-drug conjugate with a therapy that stimulates the immune system to attack tumor cells.
Health - Pharmacology - 24.11.2015
Novartis’ heart failure medicine Entresto(TM) receives EU approval
Decision brings hope of longer and better life with fewer hospitalizations for millions of Europeans with symptomatic chronic heart failure with reduced ejection fraction Entresto is the first and only treatment to show a significant mortality benefit in a head-to-head trial against ACE-inhibitor enalapril Heart failure is a life-threatening condition affecting nearly 15 million Europeans ; about half have the reduced ejection fraction form T
Pharmacology - Health - 23.11.2015
Novartis receives two landmark European approvals for Cosentyx
Cosentyx is the first and only IL-17A inhibitor from a new class of medicines shown to treat two of the most common inflammatory joint conditions in Europe Cosentyx shows rapid and sustained clinical benefits in ankylosing spondylitis (AS) and psoriatic arthritis (PsA) with no progression of spinal damage in approximately 80% of AS patients and joint damage in 84% of PsA patients as measured by x-ray over two years[2-5] Urgent unmet need for new
Health - Pharmacology - 23.11.2015

The BK polyomavirus often causes complications after kidney transplantation.
Life Sciences - Mar 27
Understanding the Brain - TU Ilmenau's EU EMBRACE Project Nominated for European Excellence Award
Understanding the Brain - TU Ilmenau's EU EMBRACE Project Nominated for European Excellence Award
Social Sciences - Mar 27
A manual addresses, for the first time in Spain, child and adolescent sexual exploitation
A manual addresses, for the first time in Spain, child and adolescent sexual exploitation

Environment - Mar 26
Changing vegetation in thawing permafrost increases emissions of greenhouse gases
Changing vegetation in thawing permafrost increases emissions of greenhouse gases

Environment - Mar 26
University of Manchester hits major sustainability milestone, with Main Campus becoming 100% 'Zero Landfill'
University of Manchester hits major sustainability milestone, with Main Campus becoming 100% 'Zero Landfill'

Social Sciences - Mar 26
"It would be naive to believe that a social media ban will solve all problems"
"It would be naive to believe that a social media ban will solve all problems"

Health - Mar 26
Earlier detection, better outcomes: Irish researchers target rising bowel cancer rates with new blood test
Earlier detection, better outcomes: Irish researchers target rising bowel cancer rates with new blood test
Environment - Mar 26
UK must improve energy efficiency to end 50 years of policy failure and prevent future energy crises, study argues
UK must improve energy efficiency to end 50 years of policy failure and prevent future energy crises, study argues

Mathematics - Mar 26
From Materials to Medical Imaging, Fonseca's Work Shapes the Future of Innovation
From Materials to Medical Imaging, Fonseca's Work Shapes the Future of Innovation










