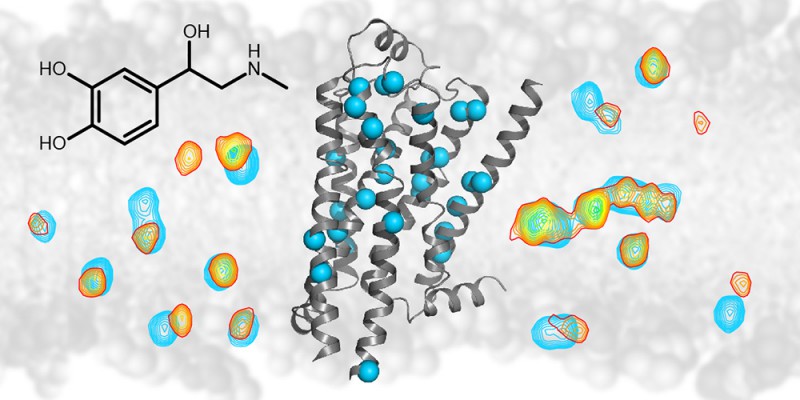
 The NMR technology detects signals (shown as contour lines) from individual atoms (blue spheres) of the ?1-adrenergic G protein coupled membrane receptor (grey ribbon diagram). Upon binding of drugs such as adrenalin (black chemical structure) the signals from the atoms change (from blue to yellow/red contours). This change allows the effect of drug binding to be followed throughout the receptor.
The NMR technology detects signals (shown as contour lines) from individual atoms (blue spheres) of the ?1-adrenergic G protein coupled membrane receptor (grey ribbon diagram). Upon binding of drugs such as adrenalin (black chemical structure) the signals from the atoms change (from blue to yellow/red contours). This change allows the effect of drug binding to be followed throughout the receptor.
About thirty percent of all medical drugs such as beta-blockers or antidepressants interact with certain types of cell surface proteins called G protein coupled receptors. Researchers at the Biozentrum of the University of Basel, in collaboration with scientists from the Paul Scherrer Institute, have now elucidated in detail how the structure of such a receptor changes when drugs bind and how the structural change transmits a signal to the cellular interior. These results have recently been published in "Nature". A wide variety of drugs such as beta-blockers against high blood pressure or drugs against allergies, cancer, Parkinson's disease, HIV and others bind to cell surface proteins which belong to the family of G protein coupled receptors. The drug binding transmits a signal to the inside of the cell. Despite the fact that many structures of these receptors are known, it remained unclear how the signal is transmitted to the intracellular inside. To better understand the signal transduction function, Prof. Stephan Grzesiek's team at the Biozentrum of the University of Basel, together with researchers from the Paul Scherrer Institute (PSI) have studied in detail one receptor - the β1-adrenergic receptor.
TO READ THIS ARTICLE, CREATE YOUR ACCOUNT
And extend your reading, free of charge and with no commitment.
Your Benefits
- Access to all content
- Receive newsmails for news and jobs
- Post ads


