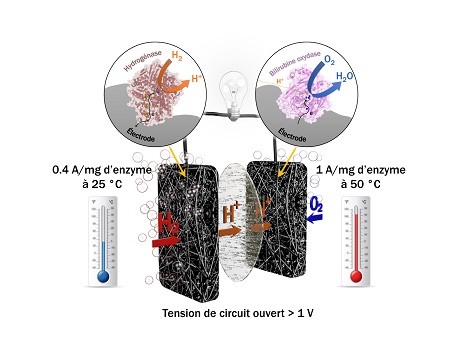
 © Ievgen Mazurenko, BIP Biocell using H2/O2 fuel based on a heat-stable hydrogenase that resists oxygen and a carbon monoxide at the anode and a heat-stable bilirubin oxidase at the cathode that provide currents of the order of amps per milligram of enzymes.
© Ievgen Mazurenko, BIP Biocell using H2/O2 fuel based on a heat-stable hydrogenase that resists oxygen and a carbon monoxide at the anode and a heat-stable bilirubin oxidase at the cathode that provide currents of the order of amps per milligram of enzymes.
Making a biocell that is as effective as a platinum fuel cell: that's the feat that researchers in the Laboratoire de Bioénergétique et Ingénierie des Protéines (CNRS/Aix-Marseille Université) have achieved, in collaboration with the Centre de Recherche Paul Pascal (CNRS/Université de Bordeaux) and the Institut Universitaire des Systèmes Thermiques Industriels (CNRS/Aix-Marseille Université). Three years after making their first prototype biocell, the researchers have just reached a new milestone and increased its performance and stability. This biocell could, in the long run, offer an alternative to fuel cells that require rare and costly metals, such as platinum. Their work was published in Energy & Environmental Science on August 17, 2017. A fuel cell converts chemical energy into electrical energy via hydrogen combustion. Though it is considered to be a clean technology because it does not emit greenhouse gases fuel cells do use costly rare metal catalysts
1, such as platinum, to oxidize hydrogen and reduce oxygen. In recent years, the identification of biocatalysts, enzymes with remarkable properties, has revitalized research in this area: their oxygen, and especially hydrogen, transformation activity is comparable to that of platinum.
TO READ THIS ARTICLE, CREATE YOUR ACCOUNT
And extend your reading, free of charge and with no commitment.
Your Benefits
- Access to all content
- Receive newsmails for news and jobs
- Post ads


