 Neural organoid with immune environment magnified twice on the left, 20 times on the right: green macrophages, red and blue neural progenitor cells (fluorescence microscopy).
Neural organoid with immune environment magnified twice on the left, 20 times on the right: green macrophages, red and blue neural progenitor cells (fluorescence microscopy).
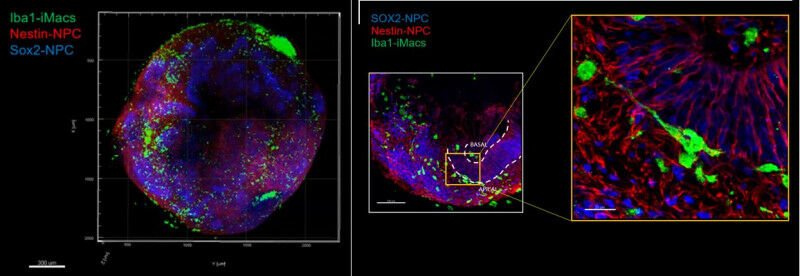
Neural organoid with immune environment magnified twice on the left, 20 times on the right: green macrophages, red and blue neural progenitor cells (fluorescence microscopy). Gustave Roussy French, Singaporean and British researchers, led by Prof. Florent Ginhoux, head of a research team at Gustave Roussy/Inserm, have succeeded in demonstrating in a neuronal organoid the role of the brain's immune environment in its formation and development. The development of these three-dimensional structures integrating neuronal cells and the immune environment is, to date, one of the most complete in vitro models of the human brain. This work is published in Nature . At Gustave Roussy, these organoids are used to model the development of childhood brain cancers, to understand their mechanisms and discover new avenues of treatment. "Although microglial cells, immune cells derived from the evolution (differentiation) of primitive macrophages present in the embryonic brain, are known to contribute to multiple aspects of brain development and function, their precise role remains poorly understood and little studied”, says Prof. Florent Ginhoux, director of a research team at Gustave Roussy/Inserm and Senior Principal Investigatorat A*STAR's Singapore Immunology Network (SIgN). The use of neuronal organoids to study their functions is one of the avenues currently favored by research.
TO READ THIS ARTICLE, CREATE YOUR ACCOUNT
And extend your reading, free of charge and with no commitment.
Your Benefits
- Access to all content
- Receive newsmails for news and jobs
- Post ads


