Researchers at the Friedrich Miescher Institute have shed new light on one of biology’s most intriguing mysteries: how identical strands of DNA give rise to such a dazzling variety of cell types, from neurons to muscle fibers.
By zeroing in on proteins called transcription factors - molecular switches that turn genes on and off - the team identified hidden patterns that determine exactly where these proteins bind along the genome and how they shape a cell’s destiny. The study, led by the Schübeler lab, reveals that these patterns form a kind of “DNA language” that influences whether a stem cell becomes part of the brain, muscle tissue, or something else entirely.
Neurons vs. Muscles: Two Factors, Two Fates
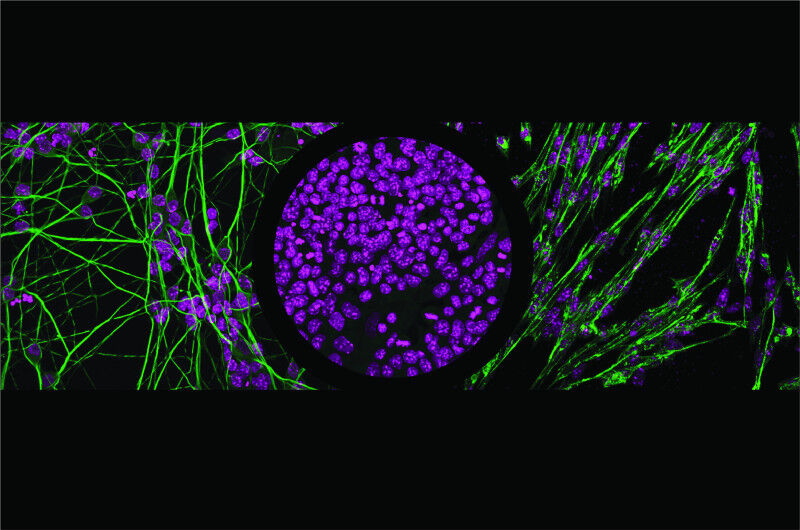
Using stem cells, the researchers focused on two closely related transcription factors: NGN2, which pushes cells toward becoming neurons, and MyoD1, which drives them toward muscle. Switching these factors on one at a time, they tracked their binding across the genome and observed the resulting changes in gene activity.
Beyond the DNA Code
The results show that binding depends on far more than just the DNA sequence. The accessibility of the DNA, the presence of partner proteins, and even tiny changes in the genetic code - sometimes just a single letter - can determine whether a gene is activated. In some cases, these transcription factors act as “pioneers,” prying open tightly packed DNA to awaken previously silent genes.
Cracking the DNA Language
To crack the binding code, the team turned to machine learning. Their model learned to predict transcription factor binding sites by recognizing specific patterns - syntax rules in the DNA language - that govern cell fate. Remarkably, the predictions held true across multiple cell types, offering a powerful new tool to anticipate, and potentially direct, the course of cellular development.
Implications for Medicine
Beyond advancing basic science, the findings could help researchers better understand developmental disorders and design strategies to influence cell fate in regenerative medicine.
Original publication
Sevi Durdu, Murat Iskar, Luke Isbel, Leslie Hoerner, Christiane Wirbelauer, Lukas Burger, Daniel Hess, Vytautas Iesmantavicius, and Dirk Schübeler Chromatin-dependent motif syntax defines differentiation trajectories Molecular Cell (2025) Advance online publication


