Unlike lizards and salamanders, whose tails can grow back, tissue regeneration is very limited in mammals. There are, however, a few well-known exceptions: deer renew their antlers every year, and rabbits are able to repair excised tissue in their ears.
Another notable exception concerns mouse embryos. A previous study [1] showed that in mice, the regeneration of forelimb buds (the front legs) is possible during the tenth day after fertilization. However, it was previously unknown how long this ability persisted and what biological and cellular mechanisms made it possible.
A new study carried out as part of the Biotherapies and bioproduction of innovative therapies research program, funded by the France 2030 investment plan and led by Inserm and CEA, now provides answers to these questions. Published in the journal PNAS, this work was led by a research team from Inserm, the University of Montpellier and Montpellier University Hospital.
our discovery shows that mouse embryos can only initiate forelimb bud regeneration during an extremely narrow developmental window: between 10.5 and 12.5 days after fertilization, when the buds are just beginning to form," explains Farida Djouad, the study’s final author. Outside this period, this ability disappears completely"
Specifically, the scientists amputated the forelimb bud of laboratory-grown mouse embryos 10.5 days after fertilization, and observed that the bud began to regenerate over the following 24 hours. However, when the same experiment was carried out 12.5 days after fertilization, no regeneration was observed.
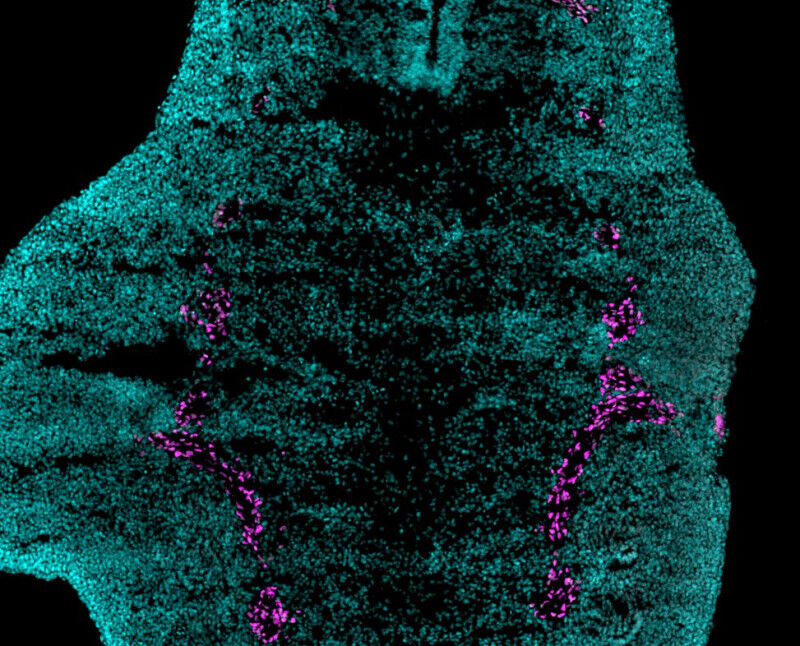
The study reveals that this ability relies on a specific population of cells called neural crest cells, known to play a key role in the development of the nervous system, facial skeleton and many tissues.
In the first three hours after amputation, these cells migrate to the injured area and help form a blastema, a cluster of immature cells that initiates regrowth. "When these cells are absent, regeneration fails. But if we transplant them again, this ability seems to be restored", stresses Farida Djouad.
Using DNA chips to analyze the activity of thousands of genes simultaneously, the scientists showed that the bmp4 and fgf8 genes, specific markers activated during limb formation from the embryonic stage, were lost after amputation but reactivated during regeneration, underlining their essential role in restoring amputated structures.
Other genes, WNT1 and FOXD3, characteristic of neural crest cells and normally active at an even earlier stage in the embryo (between days 8 and 10 post-fertilization, when limb buds begin to form), were also reactivated. " This temporary reactivation seems to return the cells to a younger, more flexible state, capable of mobilizing and participating in tissue reconstruction", continues the author.
Previous studies had already demonstrated the role of neural crest cells in the regeneration of tails and limbs in newts [2] , as well as in the regeneration of fingertips in mouse embryos [3] .
" Our results suggest that neural crest-derived cells are at the heart of regeneration mechanisms in all vertebrates, from amphibians to mammals ", explains Jholy De La Cruz, co-first author of the study.
These results offer the beginnings of an explanation for the loss of the ability to regenerate in adult mice: cells derived from the neural crest are present, but they can no longer reactivate the genes required for tissue regeneration expressed in the embryonic state.
The research team now wants to find out whether these mechanisms also exist in humans.
"Ultimately, we hope our work will contribute to a better understanding of tissue regeneration, including in humans, and how it might one day be possible to reactivate these mechanisms for therapeutic purposes," concludes Farida Djouad.
[1] Chan WY, Lee KK, Tam PP. Regenerative capacity of forelimb buds after amputation in mouse embryos at the early-organogenesis stage. J Exp Zool. 1991 Oct;260(1):74-83. doi: 10.1002/jez.1402600110. PMID: 1791423.
[2] Kumar A, Godwin JW, Gates PB, Garza-Garcia AA, Brockes JP. Molecular basis for the nerve dependence of limb regeneration in an adult vertebrate. Science. 2007 Nov 2;318(5851):772-7. . PMID: 17975060; PMCID: PMC2696928.
[3] Johnston AP, Yuzwa SA, Carr MJ, Mahmud N, Storer MA, Krause MP, Jones K, Paul S, Kaplan DR, Miller FD. Dedifferentiated Schwann Cell Precursors Secreting Paracrine Factors Are Required for Regeneration of the Mammalian Digit Tip. Cell Stem Cell. 2016 Oct 6;19(4):433-448. doi: 10.1016/j.stem.2016.06.002. Epub 2016 Jul 1. PMID: 27376984.
Neural Crest Cell Recruitment and Reprogramming as Central Drivers of Embryonic Limb Regeneration 1*BérylLaplace-Builhé, 1*GautierTejedor, 1*JholyDe La Cruz, 1AudreyBarthelaix, 2FrédéricMarmigère, 1DoraSapède, 1SarahBahraoui, 3LucieDiouloufet, 3Stéphanie Ventéo, 4JérômeCollignon, 1,5ChristianJorgensen, 1,5 Farida Djouad 1 IRMB, Univ Montpellier, INSERM, Montpellier, France; 2 IGFL, CNRS, ENS, Lyon, France; 3 INM, Univ Montpellier, INSERM, Montpellier, France; 4 Université Paris Cité, CNRS, Institut Jacques Monod, F-75013 Paris, France; 5 CHU Montpellier, Montpellier, France; * These authors contributed equally to this work.
PNAS
https://doi.org/10.1073/pnas.2519994122

